4 Types of Monoclonal Antibodies
Table of Contents
Monoclonal antibodies serve as critical tools in immunology, molecular biology, cell biology, and translational medicine. Their structural grouping plays a direct role in test dependability, immune reaction danger, approval ease, and later treatment possibilities. When picking a monoclonal antibody style, scientists need to check antigen precision, binding strength numbers, epitope spotting ways, cross-reaction, and host immune reply. They also must think about making schedules and quality control setups that guide repeat results. Beijing Solarbio Science & Technology Co., Ltd. has worked in the life science reagent area since 2004. It acts as a national high-tech enterprise integrating R&D, production, sales and service. The company has set up a full platform that covers antibodies, recombinant proteins, ELISA kits, biochemical assay kits, staining solutions, analytical standards, peptides, and small molecule compounds. The company holds ISO 9001, ISO 13485, ISO 14001, and ISO 45001 certifications. It has backed nearly 150,000 major publications, which include pieces in journals such as NATURE MEDICINE, CELL, and CELL RESEARCH. This shows the technical growth and repeat quality of its immune products.
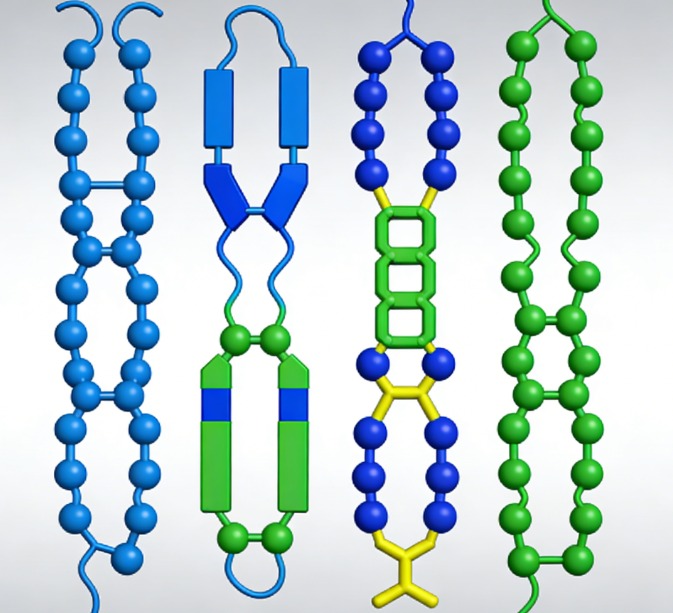
Type 1: Murine Monoclonal Antibodies
Murine monoclonal antibodies are the earliest hybridoma-derived antibody format. They are still widely used in basic research and early-stage antigen validation, owing to their high specificity and mature production protocols.
Characteristics Of Murine Monoclonal Antibodies
Murine monoclonal antibodies by immunizing BALB/c mice with purified protein or synthetic peptide antigens. Then, they harvested from the immunized mice and fused with myeloma cells (e.g., SP2/0) at a defined ratio, to generate stable hybridoma cell lines that secrete homogeneous immunoglobulin molecules . These antibodies contain complete murine variable regions (Fv) and constant regions (Fc) , which confer high binding affinity to target epitopes, including both linear and conformational epitopes, provided that appropriate adjuvants are used to maintain the native spatial conformation of the antigen.
Laboratories typically screen serum antibody titers via protein-based ELISA prior to cell fusion. Positive hybridoma lines are subjected to multiple rounds of subcloning to obtain stable monoclonal cell lines, followed by culture supernatant production and antibody affinity purification
Applications And Technical Limitations
Murine monoclonal antibodies fit well for Western blot(WB) , immunohistochemistry(IHC), immunofluorescence(IF) staining, ELISA quantification, and epitope mapping in early-stage research. However, the full murine immunoglobulin structure can induce a strong human anti-mouse antibody (HAMA) response.
Structured Development Through Custom Antibody Services
The Custom Antibody Service Platform provides end-to-end custom mouse monoclonal antibody development services, including immunization of BALB/c mice, ELISA-based serum antibody titer detection, splenocyte fusion, supernatant screening, subcloning, stable monoclonal cell line establishment, and antibody purification.
Rapid antiserum preparation can be completed within 3-4 weeks for applications where only polyclonal antiserum is required. Complete monoclonal antibody development typically takes several months, depending on the screening throughput and validation requirements.The required antigen dosage is relatively low: typically, 300-500 μg of purified antigen is sufficient for the immunization of 5 BALB/c mice, which greatly reduces the consumption of precious recombinant proteins and synthetic peptides
Type 2: Chimeric Monoclonal Antibodies
When immune reaction turns into a worry in translational research, chimeric monoclonal antibodies provide an intermediate structural format that balances high antigen-binding specificity with reduced immunogenicity risk.
Chimeric monoclonal antibodies retain the murine variable regions responsible for antigen recognition, while replacing the murine constant regions with human immunoglobulin constant regions. This design increases the overall proportion of human sequence in the antibody, thereby significantly reducing immunogenicity.
Structural Design And Immunological Considerations
The antigen-binding is located in the variable heavy (VH) and variable light (VL) chain regions. Retention of these regions fully preserves epitope specificity, while replacement of the constant regions with human-derived sequences modulates Fc-mediated immune effector functions ,and serum half-life characteristics of the antibody. Compared to full murine immunoglobulins, this structural modification significantly reduces the risk of anti-drug antibody (ADA) development
Relevance In Translational And Preclinical Research
Chimeric antibodies are widely used in receptor blocking assays, cytokine neutralization studies, and in vivo disease modelsthat require long-term antibody administration. Their partially humanized constant regions improve compatibility with human Fc receptor systems, while retaining the binding affinity to target antigens validated in the murine antibody discovery phase. This performance is further supported by a comprehensive portfolio of supporting reagents, including more than 18,000 antibody products and an extensive range of protein and immunoassay reagents, enabling integrated validation via ELISA kits, staining systems, and biochemical assay systems.
Engineering And Validation Workflow
The production of chimeric antibodies requires gene construct design, vector construction, recombinant expression in appropriate host systems, purification, and rigorous quality control (QC) validation, including ELISA-based antibody titer determination and binding specificity validation.
A systematic quality management system, covering quality planning, in-process quality monitoring, finished product acceptance inspection, root cause analysis, and continuous improvement, ensures excellent lot-to-lot consistency and regulatory approval readiness.
Type 3: Humanized Monoclonal Antibodies
Humanized monoclonal antibodies further reduce immunogenicity, as they retain only the complementarity-determining regions (CDRs) from the parental murine antibody. This format is of particular importance for therapeutic development requiring long-term administration and strict safety evaluation.
CDR Grafting And Structural Refinement
CDR grafting refers to the transplantation of antigen-binding CDR loops into a human immunoglobulin framework region (FR), while retaining key framework residues that support the correct spatial conformation of the CDRs.
In silico homology modeling and in vitro binding validation are required to avoid the loss of binding affinity in the humanized antibody framework. Routine validation assays include ELISA, Western blotting, immunohistochemistry, and cell-based functional assays, which are performed to confirm the retained binding specificity and bioactivity of the humanized antibody.
Addressing Immunogenicity And Regulatory Concerns
Humanized monoclonal antibodies typically have a human sequence content of more than 90%, which greatly reduces the risk of host immune recognition. Reduced immunogenicity improves the in vivo stability of the drug, as well as the interpretability of drug efficacy data in preclinical and clinical studies.
For in vivo applications, endotoxin control typically requires endotoxin levels below 0.3 EU/mg. Affinity purification via chromatography is a critical step in the production process to meet this requirement.
Integration With Broader Research Platforms
The company’s comprehensive life science reagent portfolio, including more than 36,000 molecular biology products, over 3,000 cell biology products, and more than 1,400 cross-species ELISA kits, provides critical support for antibody functional validation experiments.
The citation of the company’s monoclonal antibodies (including Anti-Histone H3/HIST3H3 and Anti-β-Actin antibodies) in top international journals fully demonstrates the translational reliability of its rigorously validated antibody products.
Type 4: Fully Human Monoclonal Antibodies
Fully human monoclonal antibodies represent the most advanced antibody format in terms of immune compatibility and therapeutic safety profile. These antibodies are generated using technologies including phage display libraries, humanized animal models, or single B cell sequencing, all of which produce immunoglobulins composed entirely of human sequences.
Generation Technologies And Screening Strategies
Phage display enables in vitro selection from highly diverse, large-scale antibody libraries. Humanized animal models expressing human immunoglobulin loci support the in vivo development of a complete human antibody repertoire. Single B cell sorting isolates natural human antibodies derived from the human immune response. Each of these technologies requires high-throughput screening and binding affinity characterization to select optimal antibody candidates.
End-To-End Development Ecosystem
Successful antibody development requires recombinant antigen expression, peptide synthesis for epitope mapping, biochemical assay validation, and diseased tissue staining to characterize tissue localization of the target antigen.Integrated technical support services, including primer synthesis, prokaryotic protein expression, peptide synthesis, and custom antibody development, form a seamless workflow from antigen design to validated immunoassay application.
Comparative Analysis: How To Select The Appropriate Monoclonal Antibody Type?
The selection of a monoclonal antibody format depends on the research stage, therapeutic objective, regulatory approval pathway, cost budget, and project timeline. Murine monoclonal antibodies are ideal for basic research and epitope discovery; chimeric antibodies offer reduced immunogenicity for translational research applications; humanized antibodies provide a higher safety profile for therapeutic development; and fully human antibodies are the preferred choice for applications requiring long-term administration and minimal immunogenicity.
Beijing Solarbio Science & Technology Co., Ltd. complements these strategies with a complete life science research ecosystem. Its end-to-end platforms encompass custom antibody production—including murine, chimeric, humanized, and fully human formats—alongside recombinant protein expression, peptide synthesis, ELISA kits, biochemical assay kits, and cell biology reagents. The company’s ISO-certified quality management system ensures consistent lot-to-lot reliability, high specificity, and low endotoxin levels, supporting both research and therapeutic development.
By integrating high-throughput screening, robust validation pipelines, and flexible customization services, Solarbio enables researchers to streamline monoclonal antibody workflows from antigen design to functional application. This integrated approach not only accelerates experimental timelines but also enhances reproducibility and translational confidence, making it an indispensable partner for laboratories aiming to translate antibody discoveries into clinical and diagnostic innovations.
FAQ
Q1: Which Monoclonal Antibody Type Is Recommended For Early-Stage Antigen Validation?
A: Murine monoclonal antibodies are the most recommended choice for early-stage antigen validation. This is because hybridoma technology enables high binding specificity and generates stable monoclonal cell lines that are compatible with routine assays including Western blotting, ELISA, and immunohistochemistry.
Q2: How Can Immunogenicity Be Reduced During Therapeutic Antibody Development?
A: Immunogenicity can be significantly reduced by shifting from the murine format to chimeric, humanized, or fully human monoclonal antibody formats. These formats incrementally increase the proportion of human sequence in the antibody, there by reducing the risk of anti-drug antibody (ADA) development.
Q3: What Is The Typical Timeline For Custom Mouse Monoclonal Antibody Production?
A: Custom mouse monoclonal antibody development typically includes animal immunization, serum antibody titer detection, cell fusion, subcloning, and antibody purification, and typically takes several months, depending on the screening throughput and validation requirements.
Q4: What Antigen Quantity Is Generally Required For Mouse Immunization?
A: Typically, 300-500 μg of purified antigen is sufficient for the immunization of 5 BALB/c mice in custom monoclonal antibody development projects.
Q5: Can Monoclonal Antibody Development Be Integrated With Other Research Assays?
A: Yes, monoclonal antibody development can be seamlessly integrated with supporting technologies including recombinant protein expression, ELISA kits, biochemical assay kits, and pathological staining systems, forming a complete validation workflow spanning molecular biology, cell biology, and immunology applications.