Essential Tips for Choosing the Right ELISA Kit
ตารางเนื้อหา
Selection of an appropriate ELISA kit represents a critical technical decision that directly determines the sensitivity, specificity and reproducibility of an assay, and ultimately the credibility of research outcomes. In quantitative immunoassays, subtle variations in antibody affinity, coating efficiency, enzyme conjugation stability or substrate reactivity can alter absorbance readings and introduce biases into statistical analyses. For research involving cytokine profiling, metabolic biomarker detection, pharmacodynamic assessment or translational disease model development, the use of an incompatible kit may result in repeated experimentation, sample loss and even delays in the publication of research findings.
Founded in 2004, ปักกิ่ง Solarbio Science & บริษัท เทคโนโลยี จำกัด is a national high-tech enterprise integrating the research and development, manufacturing, quality management and global distribution of life science reagents. The company has constructed a diversified technological platform and a comprehensive product portfolio encompassing antibodies, recombinant proteins, ELISA kits, biochemical assay kits, staining solutions, analytical standards and small molecule compounds. Holding ISO 9001, ISO 13485, ISO 14001 and ISO 45001 certifications, the company provides more than 100,000 in-stock products, offering full-spectrum research support for laboratories engaged in molecular biology, cell biology, immunology and other related fields. According to incomplete statistics, its products have facilitated the publication of nearly 150,000 influential research papers globally, a considerable number of which have been published in top international journals. Within its complete product line, the ELISA kit series has evolved into a well-established and fully validated standardized immunoassay system.
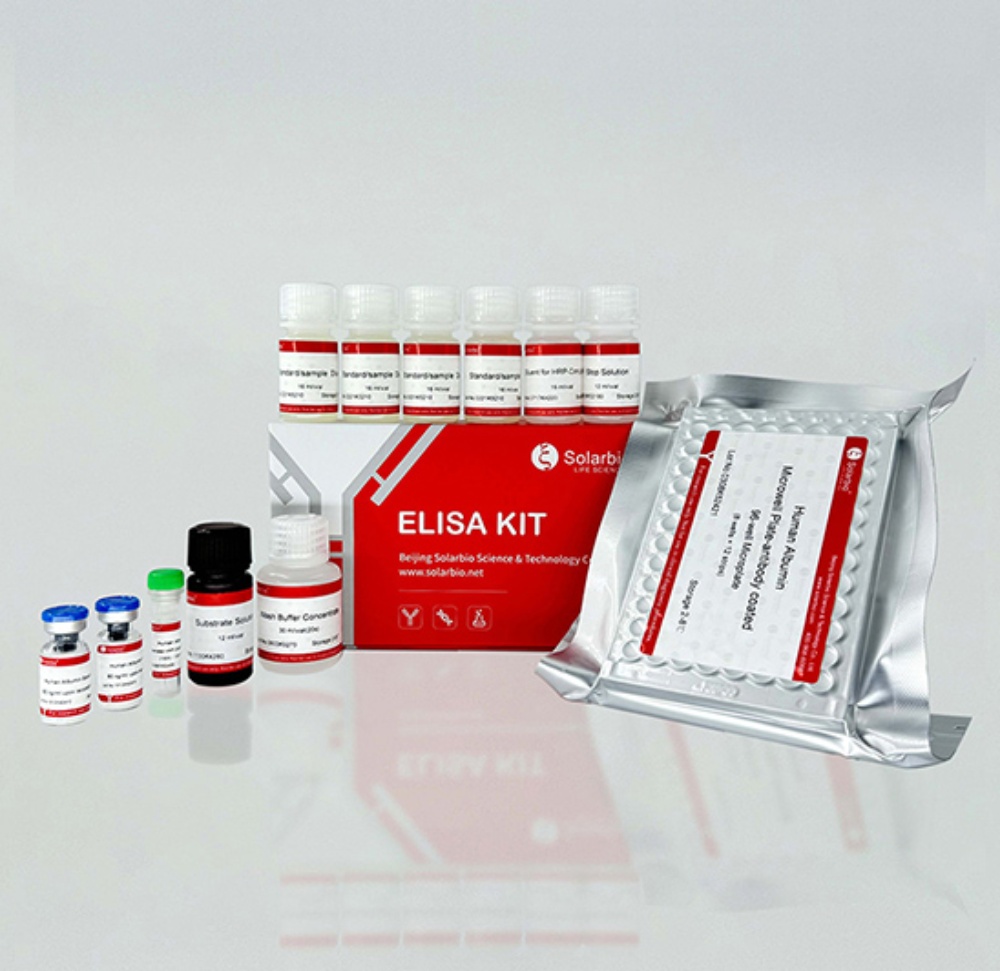
Understanding ELISA Kits: Core Principles and Scientific Value in Research
ELISA remains one of the most widely used immunoassay methods for the qualitative detection and quantitative measurement of proteins, peptides, hormones, cytokines, and viral antigens in biological fluid samples.
What Is An ELISA Kit?
An enzyme-linked immunosorbent assay relies on highly specific antigen–antibody binding. Antigens or antibodies are immobilized onto a solid support through physical adsorption while retaining their immunological activity. Detection components are conjugated to enzymes without compromising their binding or catalytic properties. Following the formation of immune complexes, the addition of a chromogenic substrate produces a detectable color change. The optical density is proportional to the analyte concentration within the validated linear range.
Solarbio provides a comprehensive ELISA product line covering sandwich ELISA, competitive ELISA, direct ELISA, one-step ELISA, high-sensitivity ELISA, and low-sample-volume formats. With more than 1,400 ELISA kits available, the product range spans multiple species including human, mouse, rat, pig, rabbit, monkey, and poultry. This extensive coverage is particularly valuable for interspecies comparative studies and translational research.
Why Rigorous Kit Selection Is Critical
Inappropriate kit selection often results in weak signal intensity, narrow detection range, high background noise, or excessive inter-assay variation. These deficiencies reduce statistical power and may obscure genuine biological differences.
Many Solarbio ELISA kits have been cited in top-tier international journals. For example, the Mouse BDNF ELISA Kit and cytokine kits targeting IL-2, IFN-γ, and TNF-α have supported studies published in journals with impact factors above 60. The Mouse Insulin ELISA Kit has been cited in CELL RESEARCH, with an impact factor exceeding 46. These citations validate the robust analytical performance of Solarbio ELISA kits under rigorous experimental conditions.
Identify Your Research Goals Before Kit Selection
A systematic kit selection process begins with a clear definition of experimental objectives. Many assay performance issues arise from misalignment between kit specifications and research purposes.
Define Quantitative Versus Qualitative Requirements
For studies requiring precise quantitation—such as biomarker quantification or drug efficacy evaluation—analytical sensitivity and dynamic range become critical parameters. In contrast, preliminary screening may tolerate greater variability.
High-sensitivity ELISA kits are ideal for low-abundance target detection, while one-step ELISA formats simplify operational procedures. Matching the assay format to research goals reduces unnecessary repetition and preserves valuable samples.
Confirm Species And Sample Compatibility
Antibody recognition of target epitopes varies across species. Therefore, species consistency verification is essential. Solarbio ELISA kits are validated for multiple biological sample types, including serum, plasma, tissue homogenates, and cell culture supernatants. This broad compatibility supports research across animal models and human sample-based studies.
Assess Sensitivity And Detection Range Needs
Sensitivity defines the minimum detectable concentration of an analyte, while dynamic range determines the full concentration interval over which reliable quantification can be achieved.
Prevent False Negative Results for Low-Abundance Targets
Cytokines and signaling molecules are often present at picogram-level concentrations. Insufficient assay sensitivity may lead to false-negative results despite genuine biological presence. High-sensitivity ELISA kits in Solarbio’s portfolio are specifically designed to address this challenge.
Auxiliary components—including high-sensitivity TMB substrate and high-affinity streptavidin-HRP conjugate—enhance color development and lower the limit of detection (LOD). Though often overlooked, these reagents strongly influence final optical density (OD) readings and assay performance.

Evaluate Standard Curve Linearity
A broad and stable linear range reduces the need for excessive sample dilution. When evaluating kits, reviewers should examine key performance metrics including correlation coefficients (R²), recovery rates, and intra-/inter-assay precision. Citations in high-impact journals further validate that the assay meets rigorous scientific standards beyond basic qualification.
Evaluate Kit Specificity And Antibody Quality
Specificity is primarily determined by antibody binding affinity and epitope recognition fidelity. Cross-reactivity introduces non-specific background signals and compromises the reliability of experimental results.
Review Antibody Production And Validation Standards
Solarbio operates a comprehensive Quality Management System compliant with ISO 9001 and ISO 13485 requirements. The system covers quality design, in-process monitoring, statistical control, root-cause analysis, and continuous improvement. This framework minimizes batch-to-batch variation and enhances consistency across production lots.
The company reports that its products were used in more than 18,000 SCI publications in 2023. This widespread adoption demonstrates stable antibody performance across diverse research scenarios.
Examine Cross–Reactivity And Interference Data
For closely related cytokines or protein families, cross-reactivity testing is critical. Thoroughly review specificity validation reports prior to kit selection. High-quality kits provide transparent data on cross-reactivity tests and recovery experiments to support confident decision-making.
Check Validation And Quality Control Documentation
Comprehensive validation documents demonstrate product development maturity and standardized manufacturing processes.
Analyze Precision And Reproducibility
Key performance indicators include intra-assay precision, inter-assay precision, and recovery consistency. Solarbio employs Acceptance Quality Level sampling and Statistical Process Control to ensure assay stability. These methodologies align with established quality criteria and minimize batch-to-batch variation.
Confirm Literature Support
When ELISA kits are cited in peer-reviewed high-impact journals such as เซลล์ และ วิจัยเซลล์, their performance validation extends beyond internal testing. Published evidence confirms reproducibility across independent laboratory settings.
Compatibility With Your Detection Platform
Confirm Microplate Reader Requirements
Most ELISA kits are compatible with standard 96-well microplate readers operating at fixed absorbance wavelengths. To ensure accurate signal detection and avoid data deviations, you must verify that your laboratory instrument fully supports the required wavelengths, temperature control, and shaking functions as specified in the kit protocol. Consistent matching between instrument settings and kit parameters prevents signal distortion and ensures reliable, reproducible data acquisition.
Verify Sample Volume Requirements
Low sample volume ELISA kits offer distinct advantages when working with limited research samples, rare animal models, or precious sample resources. A precise match between the required sample input volume and available sample availability reduces the risk of assay failure, minimizes sample consumption, and supports stable experimental outcomes even under restricted sample conditions.
Consider Kit Components And Ease Of Use
Assay workflow design directly affects laboratory efficiency and result consistency.
Confirm Complete Reagent Inclusion
A complete, ready-to-use ELISA kit should provide pre-coated microplates, calibrated standards, detection antibodies, enzyme conjugates, chromogenic substrate solution, stop solution, concentrated wash buffers, and detailed standard operating procedures (SOPs). Incomplete reagent sets introduce avoidable variability, operational delays, and inconsistent experimental outcomes.
Solarbio’s ELISA portfolio includes competitive, antibody-sandwich (ABS), one-step, and high-sensitivity formats, offering flexible experimental design without compromising analytical integrity.
Evaluate Protocol Clarity
Clear, validated, and user-friendly operating instructions reduce human error and operational variability, especially in high-throughput laboratories running parallel assays. Standardized, easy-to-follow protocols enhance reproducibility and support stable data generation across users and experiments.
Review Technical Support And After Sales Service
Assess Availability Of Professional Support
Solarbio provides professional technical consultation and comprehensive after-sales service to assist researchers throughout the experimental process. This includes targeted guidance on assay optimization, troubleshooting for abnormal signal fluctuations, and solutions to operational problems. The company has set up service centers in multiple major cities, ensuring fast response speeds and stable reagent inventory to support smooth experimental progress.
Consider Customization Options
For high-level research scenarios where standard commercial kits cannot meet specialized experimental needs, customized kit services are indispensable. Access to such personalized solutions is highly valuable for drug discovery, specific biomarker detection, novel pathway exploration and other cutting-edge research projects.
คำถามที่พบบ่อย
Q1: How Do You Choose Between Sandwich ELISA And Competitive ELISA?
A: Sandwich ELISA is typically suitable for large proteins with multiple distinct epitopes and delivers higher assay sensitivity and specificity. Competitive ELISA is ideal for small molecular targets or analytes with limited epitope sites.
Q2: Why Is Species Validation Essential In ELISA Selection?
A: Antigen-antibody binding relies on specific epitope structure, which varies across different species. Mismatched species compatibility significantly increases the risk of inaccurate quantification and false results.
Q3: What Factors Determine ELISA Sensitivity?
A: Assay sensitivity is governed by antibody binding affinity, solid-phase coating efficiency, enzyme conjugate stability, substrate catalytic reactivity, and instrument calibration accuracy.
Q4: How Do ISO Certifications Impact Reagent Reliability?
A: ISO 9001 and ISO 13485 certifications represent standardized quality management systems, which enforce consistent manufacturing processes, strict quality control and regulatory compliance, thereby guaranteeing reagent reliability.
Q5: Can ELISA Kits Support Drug Discovery And Translational Research?
A: Yes. Fully validated ELISA kits are widely applied in biomarker quantification, pharmacodynamic evaluation, and metabolic profiling for scientific research purposes only.