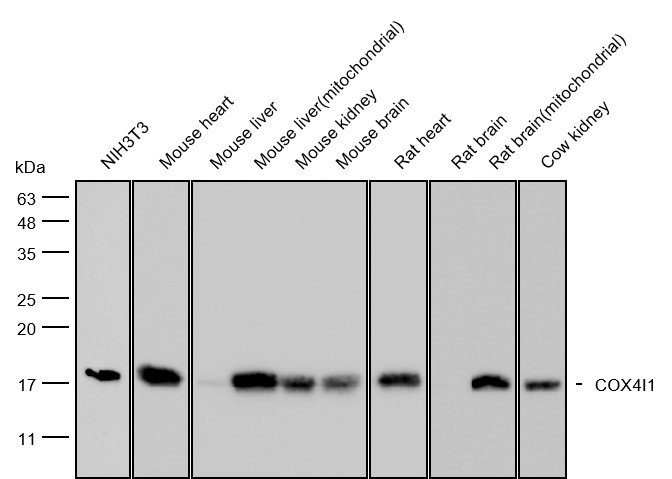
What Biological Molecule Are Antibodies?
Table of Contents
Antibodies—also known as immunoglobulins—Y-shaped glycoproteins produced by plasma cells and central to adaptive immunity. Their complex quaternary structure, combined with variable glycosylation patterns, makes them particularly sensitive to manufacturing and handling conditions. For bench scientists, this sensitivity translates into a persistent challenge: antibody lots from different suppliers—or even from the same vendor—can yield inconsistent Western blot patterns, variable ELISA signals, high nonspecific backgrounds in immunofluorescence. While rigorous manufacturing validation is essential, the root causes differ fundamentally between polyclonal antibodies (inherently variable due to polyclonal immune responses) and monoclonal or recombinant antibodies (where variability stems more from production conditions and genetic drift). Addressing this reproducibility crisis thus requires not only better quality control but also antibody validation standards that account for these biological distinctions..
Antibodies Are Specialized Glycoproteins
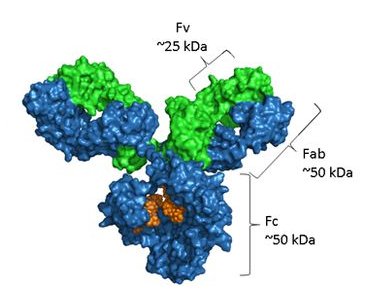
Structurally, antibodies are composed of two identical heavy chains and two identical light chains—either κ or λ isotype—linked by disulfide bonds into a characteristic Y-shaped quaternary structure . The Fab (fragment antigen-binding) regions at the arms of the Y contain variable domains (VH and VL) each bearing three complementarity-determining regions (CDRs) that confer antigen specificity. The Fc (fragment crystallizable) region mediates effector functions—such as complement activation and binding to Fc receptors on immune cells—and carries a conserved N-linked glycosylation site at Asn297 that modulates solubility, stability, and antibody-dependent cellular cytotoxicity (ADCC).
This glycoprotein architecture explains why antibody performance is exquisitely sensitive to manufacturing conditions.Because Fc glycosylation is a post-translational modification catalyzed by host-cell glycosyltransferases, its pattern is highly dependent on the expression system (e.g., CHO, NS0, or HEK293 cells) and culture parameters. IgG Fc glycans are structurally heterogeneous, and their composition directly impacts function: core fucose absence enhances ADCChrough strengthened FcγRIIIa engagement; bisecting GlcNAc and high galactose (G2) promote complement-dependent cytotoxicity (CDC); terminal α2,6-sialic acid on the Fc glycan engages DC-SIGN to exert anti-inflammatory effects; whereas agalactosylated (G0) glycoforms, prevalent in chronic inflammatory states, exhibit reduced CDC and altered Fc effector profiles. Batch variation often arises from inconsistent glycosylation during cell culture. Consequently, effective quality control must extend beyond protein concentration assays to include glycan profiling—typically by HILIC-UPLC or CE-LIF—to ensure functional consistency across manufacturing lots..
Note:Blue represents the heavy chain, green represents the light chain, and the orange part is the glycosylation.
Antibody Classification: Matching Molecular Properties to Research Needs
Understanding antibody classification at the molecular level enables informed reagent selection:
By Isotype (Heavy Chain Variants):
- IgG: Predominant serum antibody (75-80%), standard for most research applications; four subclasses (IgG1-4) with distinct complement activation capacities (IgG3 > IgG1 >> IgG2, IgG4), differential FcγR binding affinities, and unique effector profiles—notably IgG4’s anti-inflammatory properties and propensity for in vivo Fab-arm exchange
- IgM: Predominantly pentameric in serum, the first antibodyclass produced in primary immune responses; monomeric when expressed as the B cell receptor (BCR) alongside IgD
- IgA: Exists as two subclasses (IgA1, IgA2); predominantly dimeric in mucosal secretions via J-chain-mediated polymerization. IgA2’s shorter hinge region confers greater resistance to bacterial proteases, making it particularly relevant for gut microbiome research
- IgE: Monomeric, central to type I hypersensitivity and anti-helminth immunity.
- IgD: Co-expressed with IgM on naive B cells as part of the BCR; increasingly recognized for secreted functions in mucosal homeostasis and basophil-mediated immune regulation.
By Clonality:
- Monoclonal antibodies (mAbs): Derived from a single B cell clone via hybridoma technology, but now also through single B cell sorting, phage display, or transgenic animal platforms—offering unparalleled specificity for single epitopes. Ideal for quantitative assays (e.g., ELISA, flow cytometry) requiring consistent, reproducible results.
- Polyclonal antibodies (pAbs): Recognize multiple epitopes on a target antigen, providing robust signal amplification and a higher probability of detecting denatured antigens in which some—but not all—epitopes remain intact. Preferred for detecting native proteins in immunohistochemistry and when high-affinity signal amplification is critical.
By Host Species & Engineering:
The choice of host species (rabbit, mouse, goat, chicken, llama) determines epitope recognition patterns, cross-reactivity profiles, and compatibility with downstream applications. Recombinant and humanized antibodies further expand the toolkit for therapeutic and diagnostic development.
Key Biological Functions of Antibodies
As core immune effectors, antibodies perform functions mediated by distinct structural domains:
Fab-mediated functions:
- Neutralization: Block pathogen entry into host cells or neutralize toxin activity through steric hindrance and receptor competition.
- ADCC (Antibody-Dependent Cellular Cytotoxicity): Direct NK cells to lyse antibody-coated target cells via FcγRIIIa engagement..
- ADCP (Antibody-Dependent Cellular Phagocytosis): Mediate macrophage engulfment of opsonized targets through activating FcγRs
.In research, the high specificity and affinity of the antigen-binding site make antibodies indispensable detection tools for Western blot, ELISA, IHC, IF, flow cytometry, and co-immunoprecipitation (co-IP). In therapeutics, Fc-engineering strategies—such as afucosylation to enhance ADCC or mutations to silence complement activation—are actively exploited to optimize clinical outcomes.
From B Cell Biology to Consistent Manufacturing
In vivo, antibody diversity arises from V(D)J recombination and somatic hypermutation—a process essential for adaptive immunity but incompatible with the reproducibility demands of research. To overcome this inherent heterogeneity, Solarbio employs controlled platforms spanning antibody discovery to manufacturing:
Hybridoma technology: Immortalized B cell–myeloma fusions for stable, long-term monoclonal antibody secretion;
Recombinant mammalian expression: CHO cells for controlled glycosylation profiles (including engineered glycoforms via glycosyltransferase modulation) and HEK293 cells for transient production with human-like glycosylation;
Phage display: In vitro affinity maturation and selection of antibody fragments (e.g., scFv, Fab), with leads subsequently reformatted into full-length IgG for recombinant production.
Rigorous lead selection criteria—encompassing sequence-defined identity, binding specificity (validated by knockout/knockdown cell lines), thermal stability, and expression yield—are critical. Antibodies failing these benchmarks inevitably exhibit batch-to-batch variability upon scale-up, compromising experimental reproducibility regardless of the production platform employed..
The Research Pain Point: Batch-to-Batch Variability
A 2023 survey across 1,000 life-science laboratories identified reagent inconsistency as as a leading cause of reproducibility failure in immunoassays. Manifestations include weak/absent Western blot bands, high background or signal loss in IHC/IF after lot replacement, and ELISA standard curve drift. These failures originate from uncontrolled manufacturing variables—cell culture fluctuations, purification inconsistencies, uncharacterized glycoform heterogeneity—not merely insufficient validation. Without multi-application validation (WB, IHC, IF, flow cytometry, IP) coupled with specificity confirmation (e.g., knockout validation), an antibody optimized for one assay frequently underperforms in another, or exhibits unpredictable lot-to-lot inconsistency.
Solarbio Antibody Validation Framework
Beijing Solarbio Science & Technology Co., Ltd. addresses these reproducibility challenges through an integrated validation pipeline
- Multi-Application Cross-Validation
Each antibody undergoes testing across five standard applications—WB, IHC, IF, FC, and IP—plus knockout (KO) validation, matched to the target protein’s biological context.
- Affinity and Specificity Quantification
Binding strength is measured via Surface Plasmon Resonance (SPR). Cross-reactivity screening against homologous proteins ensures off-target binding is minimized.
- Lot-to-Lot Consistency Protocols
Every production batch is tracked through IPQC, SPC, and AQL evaluations. Antibodies are purified via affinity chromatography and supplied in glycerol-containing stabilizers to prevent freeze-thaw damage.
- Citation-Backed Performance
Solarbio reagents have been cited in over 150,000 SCI-indexed publications, including journals such as Nature Medicine (IF 82.9) and Cell (IF 66.85) . Specific antibody applications appear in studies ranging from rheumatoid arthritis fibroblast signaling to autophagy inhibition in cancer models, providing researchers with peer-reviewed evidence of reliability.
From Research to Reproducible Results
The classification of antibodies as glycoproteins is not merely academic—it directly informs how these reagents must be manufactured, validated, and stored to ensure experimental fidelity. By combining structural understanding with ISO-certified quality systems and multi-application validation, researchers can control for the inherent lot-to-lot variation that undermines immunoassay reliability.
FAQ
Q1: What type of biomolecule are antibodies?
A: Glycoproteins (immunoglobulins) composed of paired heavy and light polypeptide chains with N-linked glycans in the Fc region.
Q2: How do antibodies achieve high specificity?
A:Through complementarity-determining regions (CDRs) in variable domains, generated by V(D)J recombination and somatic hypermutation.
Q3: What causes antibody batch-to-batch variation?
A: Glycosylation heterogeneity, aggregation/fragmentation, and hybridoma clone drift. Rigorous QC—including glycan profiling and functional lot testing—controls for this variability.
Q4: Can antibodies be customized for novel or difficult targets?
A:Yes.Through hybridoma technology, recombinant expression, and phage display, researchers can generate antibodies against poorly immunogenic targets, (e.g., small molecules, PTMs), post-translational modifications, or conformation-specific epitopes.
Q5: Are Solarbio antibodies suitable for clinical use?
A: No. For research use only (RUO). Research antibodies are manufactured without GMP compliance, validated endotoxin limits, or regulatory dossiers required for diagnostic or therapeutic applications.
|
Cat |
product name |
Title of the literature |
IF |
|
K107441P |
Anti-MYH11 Polyclonal Antibody |
High-resolution spatially resolved proteomics of complex tissues based on microfluidics and transfer learning |
45.5 |
|
K008899P |
Anti-MYH11 Polyclonal Antibody |
High-resolution spatially resolved proteomics of complex tissues based on microfluidics and transfer learning |
45.5 |
|
K106603P |
Anti-PDIA3 Polyclonal Antibody |
High-resolution spatially resolved proteomics of complex tissues based on microfluidics and transfer learning |
45.5 |
|
K007444P |
Anti-DCN Polyclonal Antibody |
High-resolution spatially resolved proteomics of complex tissues based on microfluidics and transfer learning |
45.5 |
|
K002913P |
Anti-DCN Polyclonal Antibody |
High-resolution spatially resolved proteomics of complex tissues based on microfluidics and transfer learning |
45.5 |
|
K002098P |
Anti-PDIA3 Polyclonal Antibody |
High-resolution spatially resolved proteomics of complex tissues based on microfluidics and transfer learning |
45.5 |
|
K002095P |
Anti-MYH11 Polyclonal Antibody |
High-resolution spatially resolved proteomics of complex tissues based on microfluidics and transfer learning |
45.5 |
|
K200059M |
Anti-β-Tubulin Monoclonal Antibody |
Glibenclamide targets MDH2 to relieve aging phenotypes through metabolism-regulated epigenetic modification |
40.8 |
|
K109422P |
Anti-SLP-76 Polyclonal Antibody |
Engineering a controllable and reversible switch for CAR-based cellular immunotherapies via a genetic code expansion system |
29.5 |
|
K009635P |
Anti-β-Tubulin Polyclonal Antibody |
Implantable,Biodegradable,and Wireless Triboelectric Devices for Cancer Therapy Through Disrupting Microtubule and Actins Dynamics |
29.4 |
|
K009499P |
Anti-β-Tubulin Polyclonal Antibody |
Implantable,Biodegradable,and Wireless Triboelectric Devices for Cancer Therapy Through Disrupting Microtubule and Actins Dynamics |
29.4 |
|
K009362P |
Anti-β-tubulin Polyclonal Antibody |
Implantable,Biodegradable,and Wireless Triboelectric Devices for Cancer Therapy Through Disrupting Microtubule and Actins Dynamics |
29.4 |
|
K200059M |
Anti-β-Tubulin Monoclonal Antibody |
Implantable,Biodegradable,and Wireless Triboelectric Devices for Cancer Therapy Through Disrupting Microtubule and Actins Dynamics |
29.4 |
|
K106392P |
Anti-β-Tubulin Polyclonal antibody |
Implantable,Biodegradable,and Wireless Triboelectric Devices for Cancer Therapy Through Disrupting Microtubule and Actins Dynamics |
29.4 |
|
K008723P |
Anti-RPL13A Polyclonal Antibody |
Intrinsic targeting of host RNA by Cas13 constrains its utility |
28.1 |
|
K006299P |
Anti-LY6E Polyclonal Antibody |
Intrinsic targeting of host RNA by Cas13 constrains its utility |
28.1 |
|
K110275P |
Anti-PYCR3 Polyclonal Antibody |
Intrinsic targeting of host RNA by Cas13 constrains its utility |
28.1 |
|
K006805P |
Anti-OASL Polyclonal Antibody |
Intrinsic targeting of host RNA by Cas13 constrains its utility |
28.1 |
|
K108466P |
Anti-RBBP4 Polyclonal Antibody |
Enteric GABAergic neuron-derived γ-aminobutyric acid initiates expression of Igfbp7 to sustain ILC3 homeostasis |
27.7 |
|
K200015M |
Anti-mCherry tag Monoclonal Antibody |
Plant and animal positive-sense single-stranded RNA viruses encode small proteins important for viral infection in their negative-sense strand |
27.5 |
|
K200057M |
Anti-GAPDH Monoclonal antibody |
One-Step Symbiosis of Bimetallic Peroxides Nanoparticles to Induce Ferroptosis/Cuproptosis and Activate cGAS-STING Pathway for Enhanced Tumor Immunotherapy |
27.4 |
|
K200058M |
Anti-β-Actin Monoclonal antibody |
One-Step Symbiosis of Bimetallic Peroxides Nanoparticles to Induce Ferroptosis/Cuproptosis and Activate cGAS-STING Pathway for Enhanced Tumor Immunotherapy |
27.4 |
|
K114465P |
Anti-M6PR Polyclonal Antibody |
A Bifunctional Lysosome-Targeting Chimera Nanoplatform for Tumor-Selective Protein Degradation and Enhanced Cancer Immunotherapy |
27.4 |
|
K009721P |
Anti-RAGE Polyclonal Antibody |
Biomarker-activated multifunctional lysosome-targeting chimeras mediated selective degradation of extracellular amyloid fibrils |
23.5 |
|
K001593P |
Anti-Bax Polyclonal Antibody |
Hybrid nanogenerator based closed-loop self-powered low-level vagus nerve stimulation system for atrial fibrillation treatment |
20.577 |
|
K001594P |
Anti-BCL2 Polyclonal Antibody |
Hybrid nanogenerator based closed-loop self-powered low-level vagus nerve stimulation system for atrial fibrillation treatment |
20.577 |